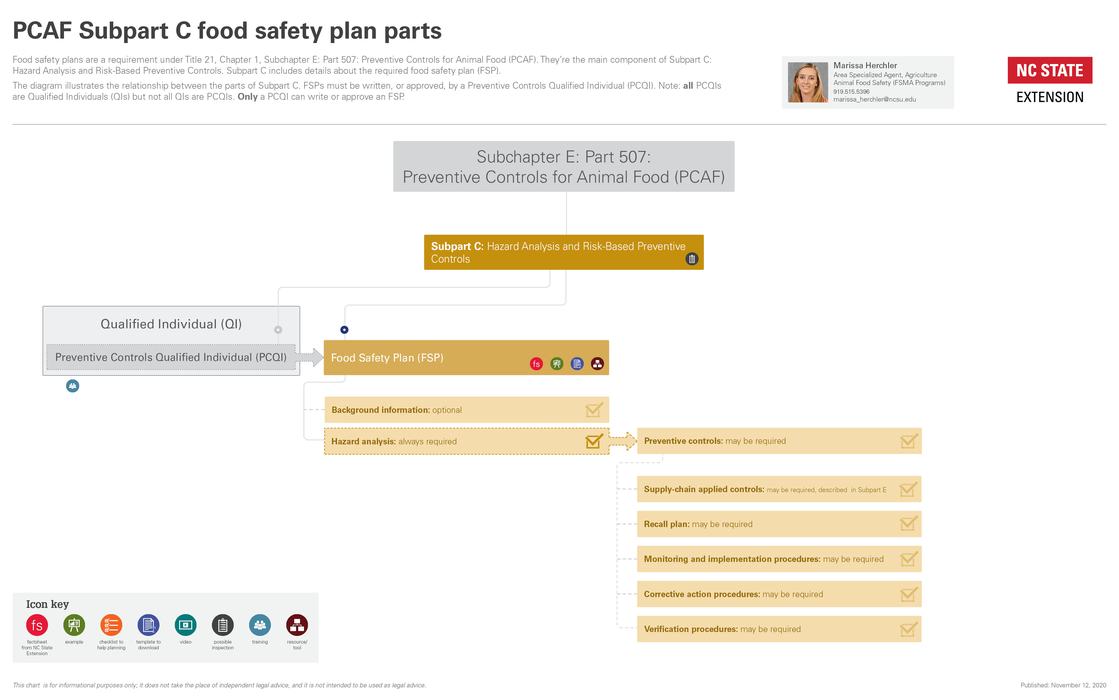
Hazard analysis is part of the regulations introduced by the FDA after passage of the Food Safety Modernization Act (FSMA) in 2011. It’s described in Subpart C of the Preventive Controls for Animal Food (PCAF) rule.
For an overview of how hazard analysis fits in with the PCAF rule, see our diagrams below.
If you are required to implement a food safety plan, a hazard analysis is always required to be part of the food safety plan.
The table below lists the management components you may need to include in your food safety plan, depending on the results of your hazard analysis.
| Management components for ensuring effectiveness of different controls | |
| Recall plan | If you identify a PC in your FSP, you must include a recall plan in your food safety plan. A recall plan is always a good business practice, whether it is needed in the food safety plan or not. |
| Monitoring and implementation procedures | Required for process PCs and sanitation PCs. |
| Corrective action procedures and corrections | If you identify a PC in your FSP, you must include corrective action procedures in your food safety plan. |
| Verification procedures | Required if a PC is identified in the hazard analysis. |
| Validation | Required for process PCs. Validation is not required for sanitation PCs because it is expected the manufacturers of sanitation solutions will validate the effectiveness of the product. |
It’s important to note that the hazard analysis is not one-size-fits-all. There are going to be differences in known or reasonably foreseeable hazards based on the species, production class and how the animals are fed. There will also be differences in preventive controls determinations, as different PCQIs will determine different severity, probability and prerequisite programs and CGMP activities per facility and based on their experiences.
There are some items that must be considered in the hazard analysis:- formulation of the animal food,
- condition, function, and design of facility and equipment,
- raw materials and other ingredients,
- transportation practices,
- manufacturing/processing procedures,
- packaging and labeling activities,
- storage and distribution,
- intended or reasonably foreseeable use,
- Sanitation, including employee hygiene, and
- other relevant factors, such as temporal (weather-related) nature of some hazards.
What is a Hazard?
A hazard, as defined by the FDA, is any biological, chemical (including radiological) or physical agent that has the potential to cause illness or injury in humans or animals.- Biological Hazards (Salmonella spp., Listeria monocytogenes),
- Chemical Hazards (mycotoxins, pesticides and process-related or industrial chemicals, drug carryover, nutrient deficiencies or toxicities), and
- Physical Hazards (stones, glass, metal).
For additional information contact
Marissa Herchler Cohen NC State University Cooperative Extension Service Prestage Department of Poultry Science 234 D Scott Hall Raleigh, NC 27695-7608Email: marissa_cohen@ncsu.edu
Phone: +1 919.515.5396